COULOMB'S LAW
An updated and better version of this article can be found here: Electrostatics and Coulomb's Law
ELECTROSTATICS
Electrostatics is that branch of Physics that deals with 'static' (constant in time) electric fields. A static electric field can only be achieved if the charges producing it are stationary.
However we will encounter some situations where the 'test charge' will be moving without the above constraints. This is because the test charge by definition is a charge which produces a negligible amount of electric field.
The reason for studying electrostatics before electrodynamics (dynamics = movement) is that a detailed study of electromagnetism is quite complicated and hence some special conditions are imposed on the subject to make it a bit easier. Once you understand electrostatics, I'll be able to confront you with more challenging aspects of electromagnetism where the restrictions of electrostatics will be removed.
The meanings of some terms like 'electromagnetism' and 'charge' that I haven't explained yet will be explained in due course. (Though I try my best to not to use any term whose meaning I have not explained but sometimes such terms are difficult to avoid. Also, I think you might know their rough meanings at least, from lower classes.)
The reason for studying electrostatics before electrodynamics (dynamics = movement) is that a detailed study of electromagnetism is quite complicated and hence some special conditions are imposed on the subject to make it a bit easier. Once you understand electrostatics, I'll be able to confront you with more challenging aspects of electromagnetism where the restrictions of electrostatics will be removed.
The meanings of some terms like 'electromagnetism' and 'charge' that I haven't explained yet will be explained in due course. (Though I try my best to not to use any term whose meaning I have not explained but sometimes such terms are difficult to avoid. Also, I think you might know their rough meanings at least, from lower classes.)
ELECTRIC CHARGE
It was known before the time of Coulomb that all massive objects attract each other by a force called gravity. Newton had given a mathematical equation to calculate such a force between two mass points.
When the electron was discovered, it was expected that two electrons (due to their masses) should attract each other by the quantitative law enunciated by Newton. However, contrary to this notion it was found that the two electrons repelled each other.
Similarly the protons were expected to attract but they repelled each other.
When the electron was discovered, it was expected that two electrons (due to their masses) should attract each other by the quantitative law enunciated by Newton. However, contrary to this notion it was found that the two electrons repelled each other.
Similarly the protons were expected to attract but they repelled each other.
The interaction between a proton and an electron was also strange. Though they attracted each other but the force of attraction between them was found to be much much larger than what was being predicted by Newton's law.
These evidences indicated that there is one more property of Nature besides gravity that is responsible for such anomalous interactions. This property was named as 'charge'.
So we can say that charge is a property of matter by which an electron repels another electron.
Note that I did not say that charge is a property by which an electron attracts a proton. Why? Because they attract each other not only due to charge but due to gravity also.
The basic definition of charge says that it is a property of matter and matter by definition is anything that has mass and occupies space. Hence it follows that charge can never be found without mass.
These evidences indicated that there is one more property of Nature besides gravity that is responsible for such anomalous interactions. This property was named as 'charge'.
So we can say that charge is a property of matter by which an electron repels another electron.
Note that I did not say that charge is a property by which an electron attracts a proton. Why? Because they attract each other not only due to charge but due to gravity also.
The basic definition of charge says that it is a property of matter and matter by definition is anything that has mass and occupies space. Hence it follows that charge can never be found without mass.
TWO KINDS OF CHARGE
Charges come in two categories which were named as positive and negative to signify their opposite nature.
It was found that:
Positive and positive charges repel each other.
Negative and negative charges repel each other.
Positive and negative charges attract each other.
To put it in one sentence:
Like charges repel while unlike charges attract.
By arbitrary convention the electron was said to possess a negative charge and the proton a positive charge.
QUANTIZATION OF CHARGE
If something occurs only in discrete quantities then we say it is quantized and the smallest discrete unit in which it is found is known as its quantum. Suppose that the minimum amount that the Indian government publishes its currency in is 1 Re. Then a person can have only 1 Re. or 2 Rs. or 3 Rs. or 100 Rs. or 1000000 Rs. etc. One cannot have 52.63 Rs. (say) because .63 Rs cannot exist. He can have 52 Rs. or 53 Rs. but not in between.
Hence Indian currency is quantized and 1 Re. is its quantum.
A similar elegant result holds for charges. The minimum amount of charge that exists in Nature is
e = 1.6 x 10^(-19) Coulomb
which is usually denoted by the letter e. A proton is found to possess this much charge. An electron too has the same charge but with a negative sign.
All other charges exist in integral multiples of this basic charge e.
Charge = ne (where n belongs to integers positive or negative).
CONSERVATION OF CHARGE
Charge can neither be created nor be destroyed. This means that the total amount of charge just after the big bang is equal to the total amount of charge at any time after it.
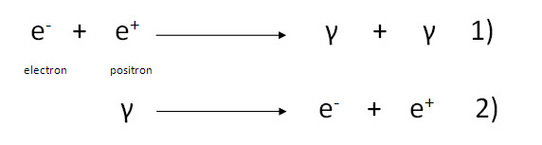
In the following two processes it seems on a first look that the conservation of charge law is being violated.
Equation 1 is known as pair annihilation while equation 2 is known as pair production.
In equation 1 an electron combines with a positron (anti particle of electron) to produce two gamma ray photons.
Equation 2 describes the situation when a gamma ray annihilates into an electron and a positron.
Equation 1 appears to have lost all charge on right hand side when it had two charges on left. Similarly equation 2 seems to have created two charges out of nowhere as its left hand side had no charge.
Well a careful look gives the following results.
The net charge on left side of equation 1 is zero since there is as much positive charge as negative charge. The charge on right is also zero. Hence no charge is lost as it didn't had any.
Similarly the right side of equation 2 has a net charge of zero since the two charges will add up to zero hence charge is again conserved here.
In equation 1 an electron combines with a positron (anti particle of electron) to produce two gamma ray photons.
Equation 2 describes the situation when a gamma ray annihilates into an electron and a positron.
Equation 1 appears to have lost all charge on right hand side when it had two charges on left. Similarly equation 2 seems to have created two charges out of nowhere as its left hand side had no charge.
Well a careful look gives the following results.
The net charge on left side of equation 1 is zero since there is as much positive charge as negative charge. The charge on right is also zero. Hence no charge is lost as it didn't had any.
Similarly the right side of equation 2 has a net charge of zero since the two charges will add up to zero hence charge is again conserved here.
COULOMB'S LAW
Just saying that like charges repel while unlike charges attract is obviously not the complete story. Two questions immediately arise:
1. What is the machinery behind this force i.e what causes the two charges to attract or repel each other?
2. What is the quantitative or mathematical law governing the force?
The answer to the first question is beyond the scope of your syllabus. Answer to the second one is described below.
After performing a series of experiments Coulomb came to the following correct conclusion. He said that the force F between any two point charges q1 and q2 separated by a distance r can be written as:
For more information on Є0 and some other constants and the relationships between them please see my blog post on
Susceptibility, Permittivity, Dielectric Constant.
Coulomb's law is valid ONLY for point charges. (A point is an entity which has a position in space but has no extent.) For larger objects, methods from integral calculus should be used to calculate the required forces.
I'll have more to say on this in next articles and in solutions to problems.
Next article is about Electric Field.
|
|
For a brilliant discussion of Electrostatics and other such topics of Physics I recommend "Fundamentals of Physics" by Resnick, Walker and Halliday.
You may buy it by clicking on the link to the left. |